Agricultural Science and Food Processing
ISSN: 3066-1579 (Online) | ISSN: 3066-1560 (Print)
Email: [email protected]


Acrylamide () is a prevalent chemical compound formed during the baking, frying, or roasting of carbohydrate-rich foods at temperatures exceeding 120°C, especially in low-moisture environments. This formation occurs through the Maillard reaction between reducing sugars (e.g., glucose, fructose) and amino acids (especially asparagine), contributing to non-enzymatic browning and imparting flavor and color to food [1, 2, 3]. Acrylamide is commonly found in bakery products such as bread, cereal, and potatoes, with the highest levels detected in roasted coffee, French fries, potato chips, and grains [4, 5, 6]. It is also used industrially as a thickener or flocculating agent in water treatment, cosmetics, and paper [7].
Despite its widespread presence and industrial applications, acrylamide poses significant health risks when present in food. It has been associated with several adverse health effects, including neurotoxicity, which can lead to nervous system damage, and reproductive toxicity, which can affect fertility and reproductive health. Additionally, acrylamide is classified as a probable human carcinogen by the International Agency for Research on Cancer (IARC), meaning it can increase the risk of developing cancer. There is also evidence suggesting that acrylamide may cause genotoxicity, which involves damage to the genetic information within a cell causing mutations [8]. The harmful effects of acrylamide were first identified in 2002 by researchers from the University of Stockholm and the Swedish National Food Administration[9]. The World Health Organization (WHO) regulates acrylamide levels in food, with daily exposure limits for adults ranging from 0.2-1.0 g/kg body weight [10].
In Europe, legislation such as recommendation 2007/331/EC monitors acrylamide levels in foods. The European Food Safety Authority (EFSA) adopted a statement in 2005, aligning with the Joint FAO/WHO Expert Committee on Food Additives (JECFA) risk assessment. A 'toolbox' guides producers on limiting acrylamide in food, with standards for nine food types across 27 EU countries. EFSA updates these recommendations based on market changes, such as the introduction of the 'other products' category in recommendation 2010/307/EC. In California, the Safe Water and Toxic Enforcement Act (1986) includes acrylamide among over 950 toxic substances, requiring companies to provide warnings if exposure levels exceed safe limits, defined as no more than one cancer case per 100,000 people over a lifetime of exposure.
In China, the Ministry of Health's 2005 Risk Assessment of Acrylamide in Food advises reducing fried and high-fat food consumption to mitigate health risks (www.gov.cn). Although specific laws are not yet in place, many large food companies have begun controlling acrylamide content, with regulations expected soon.
Acrylamide detection methods have evolved from derivatization and gas chromatography-mass spectrometry (GC-MS) [11, 12] to more advanced techniques like Liquid Chromatography-Tandem Mass Spectrometry (LC-MS/MS). Current methods include GC, High-Performance Liquid Chromatography (HPLC), and Mass Spectrometry, though these are often slow and costly. Alternatives like GC-ECD [12], HPLC with UV detection (BGVV, 2002), and reversed-phase HPLC-DAD [13] have been developed. Other methods include LC-UV [14, 15, 16], capillary electrophoresis (CE), solid-phase extraction (SPE), and dispersive liquid-liquid microextraction (DLLME) [17]. The European Committee for Standardization has published LC-ESI-MS/MS for determining acrylamide in bakery products.
Given the potential health risks, it is crucial to review and understand the sources, formation mechanisms, and mitigation strategies for acrylamide in food. This review aims to provide a comprehensive overview of current research on acrylamide, highlighting the importance of controlling its levels in food products to protect public health. By examining various detection methods, regulatory measures, and mitigation strategies, this review seeks to inform future research and policy development aimed at reducing acrylamide exposure from dietary sources.
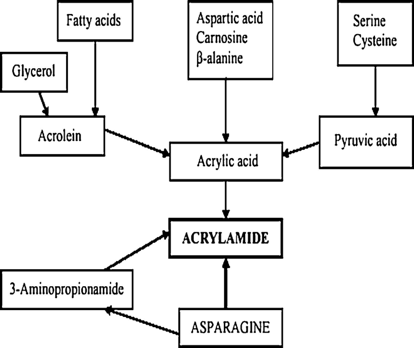
According to prior research, there are two main pathways leading to the formation of acrylamide in food. The first is the Maillard reaction, a common reaction occurring during thermal treatment. The second is fat oxidation, where acrolein released by oxidative lipid degradation leads to acrylic acid formation [18].
The Maillard reaction, occurring between reducing sugars and amino acids during food processing, is crucial for flavor, browning, and aroma development. However, it can produce toxic by-products like acrylamide, a known carcinogen [7].
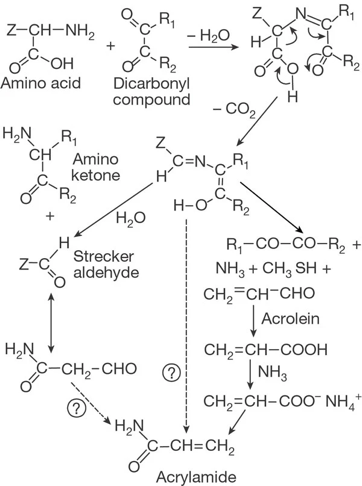
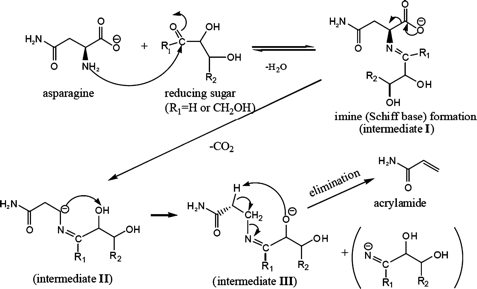
The primary pathway for acrylamide formation is the asparagine route. Asparagine and reducing sugars form Schiff bases, which decarboxylate to produce intermediates. The carboxylate ion in the Schiff base facilitates decarboxylation, leading to the removal of hydrogen from the hydroxy group and forming an alpha-hydroxy anion. This anion then removes the acidic alpha-hydrogen, resulting in acrylamide, as shown in Figure 1. The critical step is the decarboxylation of the Schiff base, leading to Maillard intermediates that release acrylamide [18].
Two main hypotheses explain this pathway. Mottram et al. [19] identified Strecker aldehyde as a key precursor, facilitated by alpha-dicarbonyls. Asparagine, abundant in potatoes and cereals, significantly influences acrylamide production, as shown in Figure 2. Stadler et al. [20] suggested that asparagine with alpha-dicarbonyls produces lower acrylamide levels compared to sugar/asparagine mixtures, but reactions with acetol yield the highest levels.
Stadler et al. [20] also proposed that early-stage Maillard reaction glycoconjugates, including N-glycosides, are pivotal in acrylamide formation. This hypothesis is supported by Yaylayan et al. [21] and Zyzak et al. [22]. Thus, while the asparagine route is primary, it involves various intermediates.
Alternative pathways exist. Figures 3 and 4 illustrate three:
Aspartic acid, carnosine, and beta-alanine enhance acrylamide formation through acrylic acid, which combines with ammonia to form acrylamide [20, 23].
Aminopropionamide, a dominant intermediate from asparagine, forms acrylamide through reactions with pyruvic acid [20, 22].
Pyruvic acid, produced by the dehydration and desulphurization of serine and cysteine, reduces to lactic acid and further dehydrates to acrylic acid, which then transforms into acrylamide [24, 25] .
Besides the Maillard reaction, fat oxidation is another pathway for acrylamide formation, usually occurring in fatty acid-rich foods as shown in Figure 4. Fatty acids first change to acrolein through oxidative degradation, and then secrete acrylic acid to achieve acrylamide formation [20, 26]. Acrolein is a common aldehyde detected as a cytotoxic component [28]. In food, it is always present in oil and fat. There is also a new pathway showing that acrolein can react with asparagine to generate high levels of acrylamide under suitable conditions, indicating a critical role of acrolein in lipid-rich foods for acrylamide formation [29, 30]. Fat, together with nitrogen compounds in food, produces acrylamide after acrolein transfers to acrylic acid. Then, there is a reaction between acrylic acid and ammonia, which can be achieved by the pyrolysis of nitrogen-containing compounds.
In industrial processes, pH plays a crucial role in many processing steps and in inhibiting the formation of certain compounds. For acrylamide (AM) formation, it is also a pH-dependent compound. Different types of food have different formation mechanisms, resulting in varying pH ranges to inhibit acrylamide.
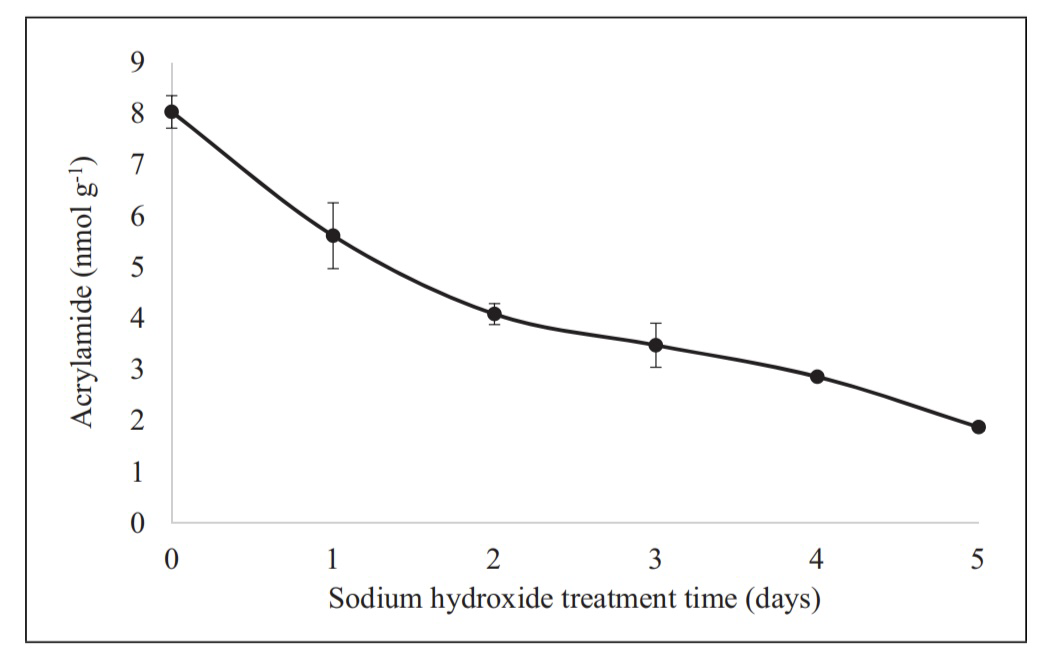
Charoenprasert et al. [31] illustrated that pH value can influence the acrylamide content in California-style black ripe olives (CBROs). Commercial olives contain high levels of acrylamide, so [31] tried to find a method to reduce these levels while preserving the olives' sensory texture. As mentioned in the formation section, acrylamide easily forms in potatoes and grains, which contain high levels of sugar. These food materials have high levels of acrylamide precursors. However, olives do not have these components, being a fatty acid-rich fruit. The exact formation mechanism in olives is still unknown, but studies suggest it is correlated with the processing step—sterilization [32]. Charoenprasert et al. [31] demonstrated that glucosamine levels before sterilization and acrylamide concentration after processing are significantly positively correlated. In their research, [31] added sodium hydroxide (, pH over 12, 0.25 N) to facilitate the base-catalyzed degradation of glucosamine. According to Table 1, the higher the pH value of the solution treating the olives, the lower the acrylamide content in the CBROs, especially at pH values above 12, with results of 3.91b ± 0.40 and 2.17c ± 0.20 at pH 12 and pH 13, respectively. Additionally, the longer the olives were soaked in sodium hydroxide, the lower the acrylamide content, provided the soaking time was within 5 days. The details are shown in Figure 5.
| Treatment solutions | Acrylamide (nmol/g)* |
|---|---|
| Control (H2O) | 8.24a ± 0.28 |
| Solution of sodium hydroxide, pH 9 | 7.79a ± 0.29 |
| Solution of sodium hydroxide, pH 10 | 8.31a ± 0.62 |
| Solution of sodium hydroxide, pH 11 | 7.93a ± 0.26 |
| Solution of sodium hydroxide, pH 12 | 3.91b ± 0.40 |
| Solution of sodium hydroxide, pH 13 | 2.17c ± 0.20 |
In the biscuit industry, pH also plays a significant role. Suman et al. [33] conducted research on factors influencing acrylamide content in wholegrain biscuits and cocoa biscuits. Biscuits are a common snack made by heating dough, which contains high levels of reducing sugars such as glucose and sucrose. Based on the mechanism, it is easy to form acrylamide in biscuits due to their carbohydrate-rich materials and high levels of precursors. In this case, pH value, heating time, and temperature are relevant factors affecting acrylamide levels. When the pH increases, the reaction between reducing sugars and asparagine is enhanced. Therefore, Suman et al. [33] emphasized the importance of adding acidification agents to biscuit dough. In the food industry, citrate and tartaric acid are commonly used as acidification agents, while biscuits need to be fluffy. This is why the authors added raising agents like sodium bicarbonate or ammonium hydrogen carbonate. Both raising agents and acidification agents help decrease acrylamide levels. Commercially, biscuits should taste good. Ensuring an acceptable range from a sensory perspective, there is a drop in acrylamide levels by more than 50% as the pH value and sodium bicarbonate concentration change. Suman et al. [33] illustrated that is more effective in limiting acrylamide formation compared to other raising agents. Graf et al. [34] determined that acrylamide levels reduce by almost 70% when sodium bicarbonate is applied.
Yuan et al. [35] showed the effects of pH value on acrylamide formation in potato chips. Although this two thermal methods (conventional heating and microwave) and the type of sugar (fructose and glucose) reacting with asparagine, their research also demonstrated higher levels of acrylamide when the pH value is high, treated by these two methods. The graph shows no acrylamide content detected when pH is equal to 3. This may relate to the form of asparagine and reducing sugars, where they present in a protonated form [36]. During the pH range from 4 to 8, acrylamide content increases in the chips. The formation rate reaches its highest when the pH value climbs to 8 in both fructose and glucose systems. This matches previous studies by Brown et al. [37] and Rydberg et al. [38], which report the maximum formation rate at pH 7 to 8. Surprisingly, acrylamide content falls after the pH rises to 10. This trend is similar to acrylamide in biscuits, as both contain high concentrations of reducing sugars.
The time of thermal treatment is critical in industrial processes, including acrylamide formation. Heating time influences acrylamide levels until a maximum point is reached [18]. The type of food also affects the required heating time.
Suman et al. [33] found that baking time significantly affects acrylamide levels in biscuits, with longer times promoting its formation, though pH is more critical than time and temperature. Deoxynivalenol (DON), a common contaminant in wholegrain bakery products, is also influenced by baking time. The greatest effect was observed when baking biscuits at 200℃ for 8 minutes. Increasing baking time effectively reduces DON contamination [39]. Reducing baking time to 5 minutes resulted in a 77-100% reduction in acrylamide levels while maintaining good sensory qualities. Thus, shorter baking times result in lower acrylamide levels in biscuits. To control both DON and acrylamide, the time-temperature parameters of baking are crucial. Baking at 180℃ for 5 minutes is optimal for cocoa and wholegrain biscuits, achieving acrylamide contents of 43±13 g/kg d.m. and 16±3 g/kg d.m., respectively, while minimizing DON content.
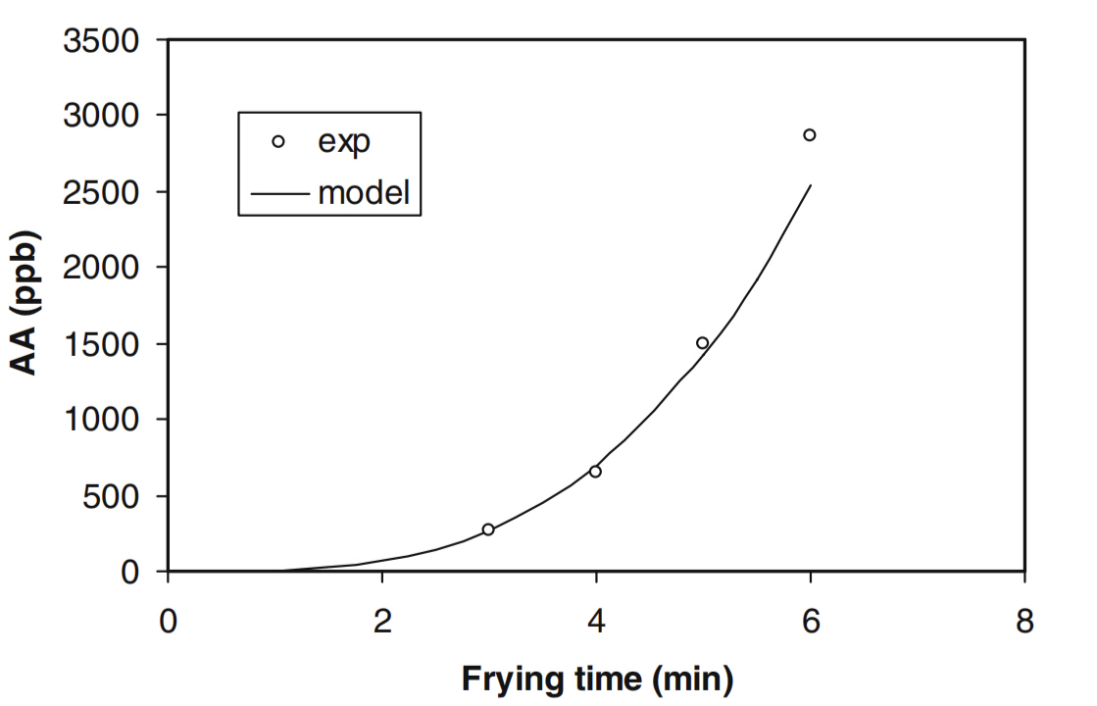
For potato chips, Tareke et al. [40] and Becalski et al. [41] demonstrated that longer frying times increase acrylamide levels. Yuan et al. [35] found that acrylamide content generally increased with longer treatment times, regardless of pH and heating methods. Acrylamide levels rose significantly after 20 minutes at pH 8.0, reaching 3.0 g/ml at 30 minutes [42]. Esposito et al. [43] recommand that control the frying time in 0-4mins to minimize acrylamide formation as shown in Figure 6.
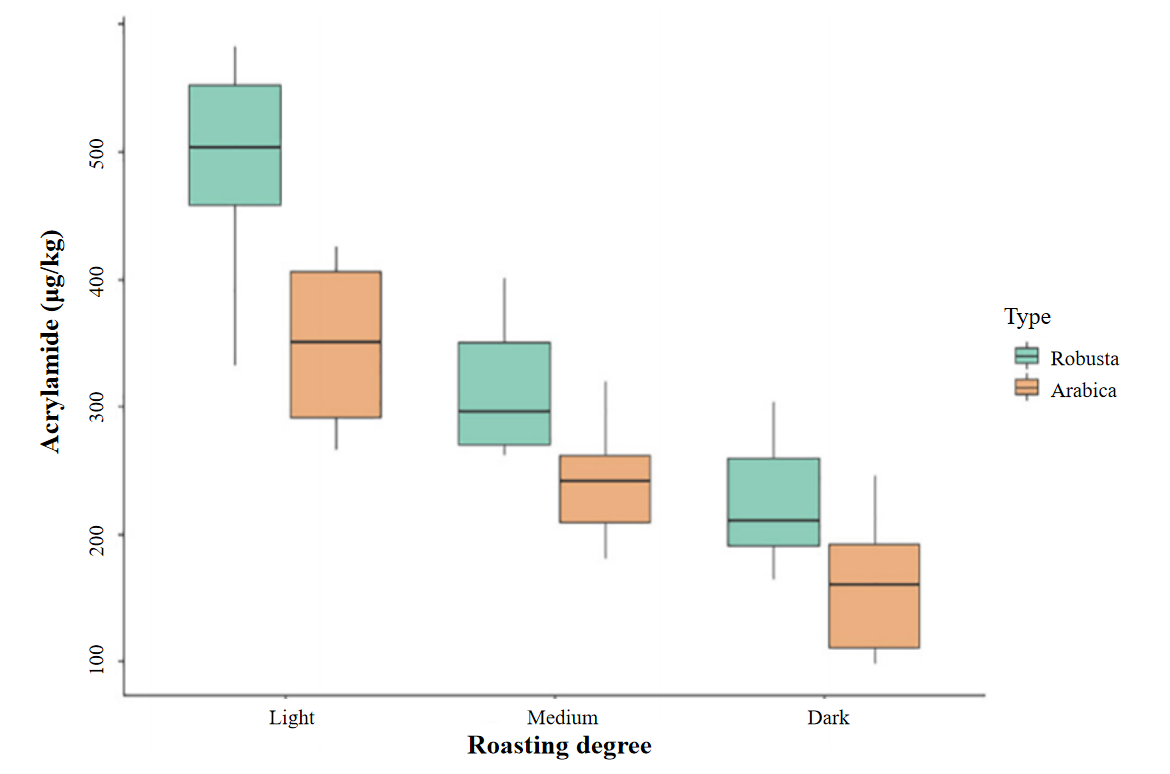
In roasted coffee, acrylamide levels are high due to precursors in coffee beans. In Figure 7, Santos et al. [44] reported that roasting at 200℃ for less than 20 minutes is optimal. Acrylamide levels peak early in the roasting process and then decrease due to degradation or depletion of precursors. Light coffee, especially Robusta, has the highest acrylamide levels ( 500 g/kg), while Arabica has the lowest ( 150 g/kg) [81, 4] .
Time also affects acrylamide levels in starch-based and cereal systems, such as bread and flatbread. Acrylamide levels peak at approximately 200℃, depending on the system and baking time [48]. Longer baking times reduce acrylamide levels, similar to dark coffee. For example, flatbread baked at 260℃ for 17.5 minutes had the lowest acrylamide levels under 20 mg/kg. Different conditions for bread crusts achieved similar acrylamide content, such as 195℃ for 19.4 or 40.6 minutes, and 230℃ for 15 minutes.
Temperature is crucial in processing, significantly influencing acrylamide levels in food. Higher temperatures in starch-based systems result in higher acrylamide content [48]. Heating temperature combined with time is critical.
In the biscuit industry, Suman et al. [33] found that baking at 200℃ increased acrylamide in wholegrain and cocoa biscuits. Reducing the temperature to 180℃ dropped acrylamide levels to below 125 ± 14 µg/kg d.m. and 156 ± 15 g/kg d.m., compared to 306 ± 16 g/kg d.m. and 400 ± 27 g/kg d.m. at 200℃. The optimal temperature for baking biscuits is 180℃ with a 5-minute baking time, achieving a 77-100% reduction while maintaining sensory quality.
For coffee roasting, Santos et al. [44] reported that roasting is usually done at around 200℃. Optimal final temperatures for different roasting levels and coffee types vary between 215-238℃ [43]. Acrylamide forms during frying, roasting, and baking above 120℃ but degrades at temperatures as low as 180℃. The effect of temperature on acrylamide levels is closely related to heating time [46, 45].
For potato chips, high temperatures lead to acrylamide formation, with reducing sugars as limiting precursors [50, 51] . Gokmen et al. [52] found acrylamide levels rise substantially with frying temperature, reaching almost 6000 ppb at 190℃ compared to 1000 ppb at 150℃. Knol et al. [47] showed that glucose converts to fructose above 160℃, and the reaction between fructose and asparagine is preferred over glucose above 140℃. High temperature and carbohydrate conversion significantly influence acrylamide formation. During frying, oil temperature above 170℃ allows temperature increase and water evaporation, favoring acrylamide formation [52].
Temperature also affects acrylamide formation in starch-based and cereal systems. Tareke et al. [40] showed acrylamide levels increase with baking temperature. Bråthen et al. [49] indicated that baked starch gel changes color from white to dark as temperature increases. Acrylamide content in freeze-dried flatbread peaks at 180℃ with a 5-minute baking time, reaching almost 3600 mg/kg. Ordinary flatbread baked at 200-220℃ achieves the highest acrylamide content. At temperatures above 260℃, acrylamide concentration peaks at nearly 500 mg/kg in bread crusts. Acrylamide reduction in starch model systems is controlled by time-temperature combinations.
To reduce acrylamide levels in food, researchers have explored various methods. These methods are compiled into a 'toolbox' to assist food companies and other researchers in acrylamide reduction efforts. This contributes to global food safety by helping residents understand the toxicity and hazards of acrylamide.
Different cooking methods are crucial in mitigating acrylamide in food, as they can produce food products that are sensory acceptable to consumers. This section introduces conventional heating methods, microwave cooking, and air-frying. Conventional heating methods, such as boiling, frying, steaming, and baking, have been used for many years. However, they do not significantly reduce acrylamide levels in food. For instance, the acrylamide content in roasted coffee powder ranges from 135 g/kg to 1139 g/kg, while French fries contain 36 g/kg to 1441 g/kg, and potato chips range from 211 g/kg to 3515 g/kg [53]. Boiling provides the lowest acrylamide levels in food. At a pH of 8.0, the optimal condition for acrylamide formation, the acrylamide content in potato chips ranges from 0.5 g/ml to 0.9 g/ml, which is lower than when using microwaves.
Microwave cooking, developed by Raytheon engineer Percy Spencer in 1945, is a widely used thermal method. Yuan et al. [35] found that microwave ovens can also be used in potato chip production. Under the same pH conditions, lower microwave power results in less acrylamide formation. This reduction can be monitored by controlling the microwave power. Compared to traditional frying (180°C for 4 minutes), microwaving at 650W for 3 minutes reduces acrylamide levels by 100 g/kg. Sansano et al. [54] reported similar findings, with acrylamide levels decreasing when microwaving at 600W for 1 to 5 minutes. For instance, the acrylamide level is 63(5) g/kg using deep-oil frying for 2 minutes, but decreases to 25(5) g/kg with microwave frying. The formation of acrylamide by microwave frying appears to depend on the frying technique. Except at the end of frying, the acrylamide content of potatoes is higher than that of microwave-fried potatoes. During the microwave process, acrylamide levels increase exponentially in potato products [54]. However, some studies found that microwaving might favor acrylamide formation in some frozen par-fried French fries [55]. Therefore, specific conditions are necessary to effectively reduce acrylamide content using microwaves.
Air-frying is a recent cooking technique that has gained popularity in households. It is a small countertop convection oven designed to simulate deep frying without adding extra oil. It can be used to bake cakes, make French fries or potato chips, and roast chicken wings. Sansano et al. [56] conducted a study comparing air-frying and deep-oil frying at 180°C. The results show that, although the Maillard reaction rate is faster for deep-frying, air-frying reduces acrylamide levels by 90% without any pretreatment in fried potatoes. This is due to differences in heat transfer between the two thermal technologies. With both mediums, oil and air, at 180°C, oil is more likely to conduct heat to the potato surface [57]. In [54]'s studies, the CIE Lab* parameters were used to present acrylamide content, as the color of the potato changes with increasing acrylamide content. Frying technology most affected the variation of color parameters during cooking, especially a*. The L* value of air-frying remains almost constant, while deep-oil frying results in darker potatoes. The b* value indicates that deep-oil fried potatoes are more yellow than air-fried ones. Thus, air-frying can be used instead of traditional oil frying to reduce acrylamide levels in food products, specifically potato chips or French fries.
Vacuum frying is a new technique involving deep-frying and dehydrating food at low temperatures (80-120°C), which effectively reduces nutrient damage caused by high temperatures. Garayo et al. [58] reported that vacuum frying could be an alternative method to produce potato chips with lower acrylamide and oil content, and better texture. The study used temperatures of 118°C, 125°C, and 140°C at a vacuum pressure of 10 Torr. Acrylamide levels decreased by 63% from 140°C to 125°C and by 10% from 125°C to 118°C. Compared to traditional frying, vacuum frying at 118°C reduces acrylamide content in potato slices, resulting in golden color and satisfactory texture. The highest acrylamide content by vacuum frying is over 500 ppb, while traditional frying results in almost 800 ppb. The CIELab* parameters show that vacuum-fried potato chips have lower acrylamide levels than traditionally fried chips [59, 60, 61]. Overall, vacuum frying can replace traditional oil frying for making potato chips.
Among the cooking methods for preventing acrylamide formation, air-frying is the most effective. Studies have shown that air-frying can reduce acrylamide levels in fried potatoes by up to 90% without any pretreatment [56]. Compared to traditional oil frying, air-frying uses hot air to cook food, which reduces the likelihood of high temperatures that promote acrylamide formation while maintaining food quality and sensory properties [57]. Additionally, air-frying reduces the overall fat content in food, making it a healthier cooking method. Therefore, air-frying is recommended as the best cooking method for reducing acrylamide formation.
Asparaginase is an enzyme present in living organisms, plants, and animals [62]. It hydrolyzes asparagine to aspartic acid and ammonia [63], reducing acrylamide formation by decreasing asparagine levels. The enzyme is derived from sources like E. coli, A. oryzae, and A. niger. Researchers have used asparaginase to reduce acrylamide in French fries, bread, potato chips, and biscuits. Zyzak et al. [22] used E. coli asparaginase, achieving a 99% reduction in acrylamide in potatoes. Ciesarová et al. [64] treated potatoes at 180°C for 20 minutes, resulting in a 50-90% reduction. Mahajan et al. [65] used Bacillus licheniformis, achieving an 80% reduction. For French fries, Pedreschi et al. [66] used A. oryzae, achieving a 67% reduction at 175°C for 3 minutes. Zuo et al. [67] used Thermococcus zilligii, achieving an 80% reduction at 175°C for 5 minutes. For potato chips, Hendriksen et al. [63] showed that 10,500 ASNU/L of asparaginase reduced acrylamide from 1700 to 700 g/kg (60% reduction). Pedreschi et al. [68] achieved a 90% reduction at 170°C for 5 minutes. Onishi et al. [69] used Bacillus subtilis, achieving an 80% reduction. For bread, Tuncel et al. [70] used Pisum sativum L., achieving a 57-68% reduction at 220°C for 22-25 minutes. Ciesarová et al. [71] used A. niger, achieving up to a 90% reduction. Kumar et al. [72] used Cladosporium sp., achieving a 97% reduction in crust and 73% in crumbs at 220°C for 25 minutes. Amrein et al. [73] used E. coli, achieving a 55% reduction in gingerbread. Huang et al. [74]used Rhizomucor miehei, achieving a 94.2% reduction in biscuits at 200°C for 25 minutes. Anese et al. [75] used different enzyme dosages in biscuits, achieving a 69% reduction at 200°C. Hendriksen et al. [63] used A. oryzae, achieving a 65-84% reduction in semi-sweet biscuits and 34-90% in ginger biscuits. The optimal conditions were pH 7.0 and 40°C, with 1050 ASNU/kg of enzyme reducing acrylamide to 10 g/kg compared to 450 g/kg in controls.
As mentioned, acrylamide formation is strongly related to reducing sugars. Researchers have indicated that reducing sugar concentration plays a key role in acrylamide formation. Therefore, decreasing reducing sugar levels should lower acrylamide content. The type of reducing sugar (glucose, fructose, sucrose) also affects acrylamide levels in carbohydrate-rich foods.
In industrial biscuit production, Suman et al. [33] reported that glucose significantly increases acrylamide levels. Using high dextrose levels at 200°C for 8 minutes increased acrylamide by 120% compared to controls. Conversely, a low glucose concentration combined with suitable time-temperature conditions (180°C, 8 minutes) resulted in a 77% decrease in acrylamide. Using sucrose as the reducing sugar source in biscuits can decrease acrylamide production by about 50% [34, 76]. Graf et al. [34] noted that low acrylamide levels form at 160°C, remaining below 150 µg/kg, and recommended using sucrose at 180°C for biscuit production.
In the potato products industry, Yuan et al. [35] found that acrylamide levels were lower when using glucose compared to asparagine-fructose combinations. At pH 8.0, the optimal condition for acrylamide formation, the acrylamide level was 1.0 µg/ml for Asn & Fru boiled at 120°C for 30 minutes, while it was 0.52 µg/ml for Asn & Glc combinations. At pH 3.0, acrylamide generation was almost zero, regardless of the combination. Muttucumaru et al. [77] showed that the ratio of asparagine to reducing sugar is crucial for acrylamide formation in potato products, with a tipping point value of 2.257 ± 0.149. Potatoes in Doncaster had higher free asparagine levels than in Woburn, resulting in lower average acrylamide levels among 20 types. Reducing sugar levels increased with storage time, but acrylamide levels were not highest in Harmony potatoes due to their lower asparagine-to-reducing sugar ratio.
Liyanage et al. [78] found that the percent decrease in asparagine content (1–60%) was generally lower than that in reducing sugars (28–94%) with increasing frying time at a constant temperature. Amrein et al. [50] reported that acrylamide formation is primarily limited by available reducing sugars rather than free asparagine. Elmore et al. [79] found that only 0.29% of asparagine was converted to acrylamide at 180°C, while reducing sugars were dominant in acrylamide formation. Bread contains various reducing sugars, including glucose, fructose, and maltose, which contribute to acrylamide formation. Crawford et al. [80] identified sugar as a factor in acrylamide formation in commercial flatbread but lacked accurate data for analysis.
In coffee products, different coffee beans contain varying percentages of asparagine and reducing sugars, resulting in different acrylamide levels. Arabica coffee has lower acrylamide levels than Robusta due to its lower asparagine content at similar roasting degrees [43, 81].
Pretreatment is a crucial step before processing, involving a series of cleaning operations to remove impurities that adversely affect dyeing and printing. For example, soaking materials in a solution or adding a solution to the materials can be beneficial. Pre-thawing frozen materials to change their form is also a type of pretreatment. Various pretreatment methods are used to reduce acrylamide levels, depending on the type of food. For instance, significant research has focused on potato products.
Microwave pre-thawing is a method used to thaw frozen food. Due to commercial demands, many potato strips are frozen. Some authors suggest that pre-thawing frozen potato strips to decrease frying time may be an effective approach to reduce acrylamide levels [82]. The authors fried potato slices at 170°C, 180°C, and 190°C after pre-thawing. The acrylamide level dropped by 10% after pre-thawing the potato slices, from 17.7 to 15.9 ng/g. The researchers recommend frying at 180°C after pretreatment, as this can reduce acrylamide levels by 89%, a significant decrease, from 72.1 to 8 ng/g. When frying at 190°C, the reduction in acrylamide was 64%, from 50.5 to 18.4 ng/g. The combination of pre-thawing and heating temperature is crucial for acrylamide reduction. When frying at 170°C, the acrylamide level showed a slight decrease, possibly because acrylamide formation is not accelerated at this temperature. Previous studies indicated that the acrylamide formation rate increases significantly above 175°C [26]. This study also examined the quality of the final products after treatment and frying. Lab* data showed that all control samples were slightly more yellow than the pre-thawed ones, which may be more appealing to consumers. All data were compared with control samples fried at the same temperature without pre-thawing.
Hydrocolloids are polysaccharides connected by glycosidic bonds, consisting of more than ten monosaccharides. Polysaccharides can be classified as homo- and hetero-polysaccharides. Homo-polysaccharides consist of identical monosaccharides, such as cellulose and amylose. In contrast, hetero-polysaccharides consist of different monosaccharides (at least two types), including alginate and xanthan. Hydrocolloids have a high affinity for aqueous solutions and are used in various industries. They can function as thickeners, gelling agents, foam stabilizers, emulsifiers, and dispersants, inhibit the formation of ice and sugar crystals, and control the release of flavors.
In 2020, Liu et al. [83] demonstrated that using sodium alginate as a coating for fried potato chips can reduce acrylamide levels in food products. Alginate is a polysaccharide extracted from seaweed. After acidification, alginic acid, which is soluble in or , is converted to sodium alginate, an anionic polymer. Liu et al. [83] determined that a sodium alginate concentration of 1.34%, frying at 179°C for 4.38 minutes, was optimal. The evidence showed that sodium alginate significantly reduced acrylamide levels, inhibiting up to 76.59% compared to control samples. The sodium alginate coating effectively prevented potato chips from absorbing oil while maintaining good quality and sensory properties. Due to the poor solubility of sodium alginate, researchers used low dosages ranging from 0.2% to 2.2%. They found that around 1.4% sodium alginate provided the best inhibition rate, reaching 58.93%. Sodium alginate contains M-blocks and G-blocks, both contributing to the viscosity and thickening of products, which explains the reduced oil absorption. Sensory evaluations indicated that coated potato chips tasted similar to regular ones. Sodium alginate also enhances satiety signals to the brain, reducing blood cholesterol, glucose levels, and total daily calorie intake. Data from several studies suggest that other hydrocolloids, such as guar gum and pectin, also reduce oil uptake during frying [84, 85]. However, sodium alginate is the most effective coating.
Champrasert et al. [86] reported that alginate, pectin, and chitosan positively affect acrylamide reduction. Pectin is a complex group of plant polysaccharides, consisting of galacturonic acid, rhamnose, arabinose, xylose, and other sugars, commonly added to fruit juice or jelly depending on the esterification of galacturonate residues. Chitosan, a linear polysaccharide, consists of glucosamine and GlcNAc randomly distributed and connected through -glycosidic bonds. In this study, polysaccharides were used as pretreatment solutions for soaking potatoes. The potato products were fried at 180°C or microwaved at 800W for 60 seconds. Under conventional heating, the inhibition rate of acrylamide was effective, at 65% for alginate and 56% for pectin. However, the reduction decreased under microwave heating, to 36% and 30%, respectively, as high-efficiency microwaves may promote acrylamide formation. Additionally, Champrasert et al. [86] coated potatoes with alginate, pectin, and chitosan at the same concentration, frying at 170°C for 3 minutes and microwaving at 800W. The reduction of acrylamide was 53.5%, 51.2%, and 40.9%, respectively, when frying. When microwaved for 60 seconds at 800W, the reduction was slight, only 5%, with no reduction for chitosan-coated potatoes. Microwave irradiation may destroy the coating, preventing it from avoiding the temperature increase that forms acrylamide.
Sansano et al. [56] demonstrated that various chemical agents can be used as pretreatment solutions to reduce acrylamide levels, such as citrate, glycine, calcium lactate, , or nicotinic acid (Vitamin B3). The authors controlled the conditions under both air-frying and deep-oil-frying at 180°C. For oil-frying, the most effective method was soaking in 2% citric acid, which reduced acrylamide levels by almost 91% (from 125 g/kg to 11 g/kg) compared to the control sample. For air-frying, 2% calcium lactate was the most effective pretreatment solution, providing a 75% decrease in acrylamide levels (from 29 g/kg to 7 g/kg). The reduction is attributed to the decrease in reducing sugars after pretreatment. Interestingly, some pretreatments did not help reduce acrylamide levels. For example, the acrylamide level in samples treated with 1% was 50% higher in air-frying compared to the control sample. This phenomenon is due to an increase in reducing sugar content after treatment with 1% , from 0.20(0.03)d to 0.21(0.02)d. Similar results were observed in samples pretreated with 1% sodium chloride solution, blanching, soaking in 1% or 2% citric acid, 2% , and 1% calcium lactate when using air-frying. , commonly known as salt, is an ionic compound, and calcium lactate is a salt consisting of two lactate anions with each cation. Another reason for the increase in acrylamide levels is that the presence of these mono- or divalent cations (Ca2+ or Na+) on the product surface could reduce the water activity of the surface, favoring acrylamide formation in the fried product. However, in traditional oil-frying, as long as the potato strips were pretreated, the chips had lower acrylamide levels than the control samples.
Lactic acid bacteria (LAB) are Gram-positive, low GC, acid-tolerant, mostly non-sporulating, non-respiratory bacilli or cocci. Found in decomposing plants and dairy products, LAB primarily produce lactic acid from sugar fermentation, historically used to increase acidity and prevent spoilage in food fermentations. Bread, a carbohydrate-rich food, requires fermentation, and several studies have investigated LAB starters to reduce acrylamide levels.
Bartkiene et al. [87] demonstrated that different LAB strains (P. acidilactici, L. plantarum, L. curvatus, and L(+) lactic acid bacteria) grown at 30°C and 37°C showed varying acrylamide reductions. Dastmalchi et al. [88], and acrylamide formation in bread is influenced by many factors, especially reducing sugars [89]. However, L. plantarum significantly reduced acrylamide concentrations in bread samples. Whole rye flour, rich in cell walls, undergoes hydrolysis and solubilization during sourdough fermentation, facilitated by LAB enzymes. L. plantarum exhibited the lowest pH and highest amylolytic enzyme activity, likely contributing to the greatest acrylamide reduction (12 g/kg dw) compared to the control (15 g/kg dw).
Bartkiene et al. [87] also reported that LAB combinations can reduce acrylamide in bread. The most effective combination, L. plantarum and P. pentosaceus, reduced acrylamide levels to 4.01±0.31 g/kg, with the lowest level at 3.70 g/kg in wheat bread. Additionally, this combination had the lowest a* value among all samples.
This article summarizes the sources of acrylamide formation and preventive measures. Acrylamide is a toxic compound that may have carcinogenic effects on the human body. It is mainly produced through the Maillard reaction between sugars and amino acids. To protect public health, governments and international organizations have enacted relevant regulations to limit the content of acrylamide in food.
The article points out that future research should focus on finding economical and widely applicable methods to reduce acrylamide content, while controlling key production steps. For example, exploring the effects of using colloids in foods that are prone to acrylamide formation (such as coffee and bread), or studying whether pre-treatment of bread can reduce the acrylamide content after baking. In addition, extracting acrylamide from food for packaging or other industrial uses may also be a future research direction, although there are still certain challenges.
In summary, by controlling key factors such as temperature, time, and pH, as well as adopting strategies like enzyme treatment, alternative cooking methods, and reducing carbohydrate content, the acrylamide content in food can be effectively reduced to protect consumer health. Future research should further explore more economical and efficient methods to reduce acrylamide.
 Copyright © 2025 by the Author(s). Published by Institute of Emerging and Computer Engineers. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/), which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made.
Copyright © 2025 by the Author(s). Published by Institute of Emerging and Computer Engineers. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/), which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. Agricultural Science and Food Processing
ISSN: 3066-1579 (Online) | ISSN: 3066-1560 (Print)
Email: [email protected]

Portico
All published articles are preserved here permanently:
https://www.portico.org/publishers/iece/