Abstract
This study investigates the impact of three precipitation methods, ammonium sulfate (40%), acetone, and acetone with trichloroacetic acid (TCA) on the yield and activity
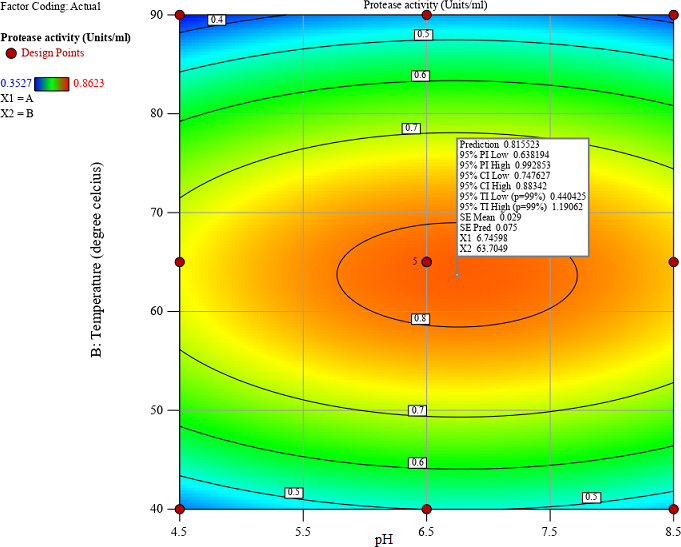
of protease extracted from Averrhoa carambola (starfruit) at various maturity stages (unripe, semi-ripe, and ripe). The study employed response surface methodology (RSM) to optimize hydrolysis conditions, specifically temperature, and pH, for maximum proteolytic activity. The proteases were incubated in buffers at pH values ranging from 3.5 to 8.5 for 12 hours, and the proteolytic activity was assessed. Additionally, temperature effects were evaluated by incubating proteases at temperatures between 40◦C and 90◦C. The storage stability of the proteases was monitored over a 7-day period at temperatures <4◦C. The results revealed that ammonium sulfate precipitation (40%) yielded the highest proteolytic activity (PA), particularly at the unripe stage, where it reached 0.34 units. Conversely, acetone with TCA precipitation exhibited the highest specific activity, with values of up to 2.38 U/mg at the ripe stage, indicating that although the total protease yield was lower, the enzymes extracted were more efficient per unit of protein. Acetone precipitation showed intermediate values for both PA and specific activity. Numerical optimization identified the optimal conditions for proteolytic activity as 65◦C and pH 6.5, achieving a maximum activity of 0.862 units/mL. Despite the promising proteolytic activity under optimized conditions, a significant reduction in enzyme activity was observed during a 7-day storage period at <4◦C. This finding underscores the importance of considering both total proteolytic activity and specific activity when selecting the optimal protease extraction method. Overall, this study demonstrates that ammonium sulfate precipitation is the most effective method for extracting protease from starfruit, particularly at the unripe stage for higher yield, while acetone with TCA precipitation offers advantages in producing more active proteases, suitable for applications requiring enzymatic efficiency.
Data Availability Statement
Data will be made available on request.
Funding
This work was supported without any funding.
Conflicts of Interest
The authors declare no conflicts of interest.
Ethical Approval and Consent to Participate
Not applicable.
Cite This Article
APA Style
Adhikari, N., Paudel, R., & Maskey, B. (2025). Effects of Precipitation Methods on Protease Yield and Proteolytic Activities of Protease Enzyme from Different Maturity Stages of Starfruit (Averrhoa Carambola). Agricultural Science and Food Processing, 2(1), 56–67. https://doi.org/10.62762/ASFP.2025.993704
Publisher's Note
IECE stays neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions

Copyright © 2025 by the Author(s). Published by Institute of Emerging and Computer Engineers. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (
https://creativecommons.org/licenses/by/4.0/), which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made.


 Submit Manuscript
Edit a Special Issue
Submit Manuscript
Edit a Special Issue
 Copyright © 2025 by the Author(s). Published by Institute of Emerging and Computer Engineers. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/), which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made.
Copyright © 2025 by the Author(s). Published by Institute of Emerging and Computer Engineers. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/), which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. 