Agricultural Science and Food Processing
ISSN: 3066-1579 (Online) | ISSN: 3066-1560 (Print)
Email: xucm@btbu.edu.cn


 Submit Manuscript
Edit a Special Issue
Submit Manuscript
Edit a Special Issue
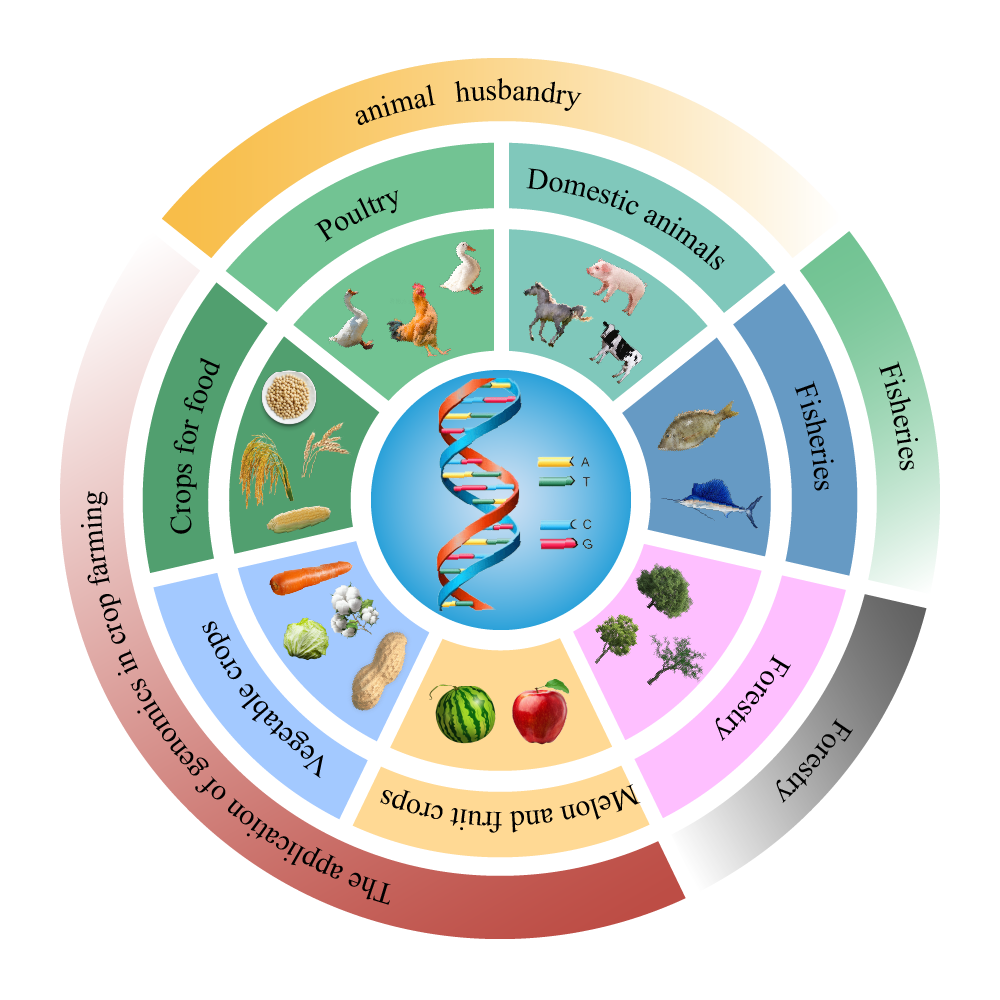
 Copyright © 2025 by the Author(s). Published by Institute of Emerging and Computer Engineers. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/), which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made.
Copyright © 2025 by the Author(s). Published by Institute of Emerging and Computer Engineers. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/), which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. Agricultural Science and Food Processing
ISSN: 3066-1579 (Online) | ISSN: 3066-1560 (Print)
Email: xucm@btbu.edu.cn

Portico
All published articles are preserved here permanently:
https://www.portico.org/publishers/iece/